 
As a cheap and reliable method of determining state of charge, the hydrometer also reveals differences between cells and allows visual inspection of the electrolyte colour. All non-sealed batteries should be checked using a hydrometer. 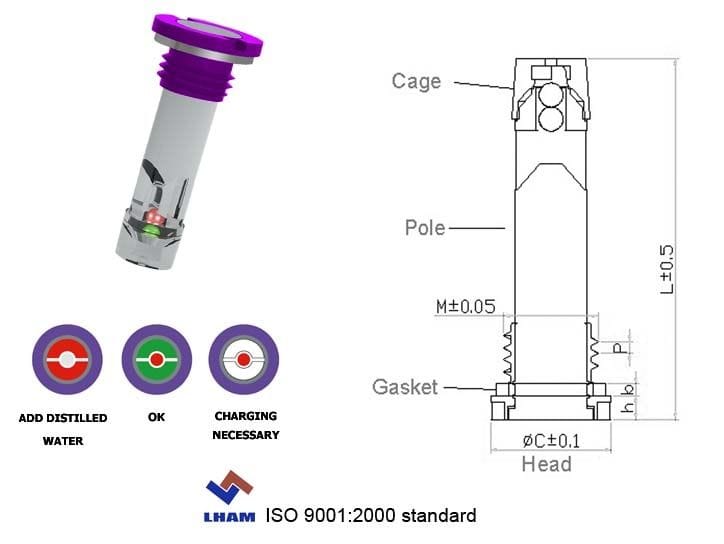
The first step in evaluating starting capability involves testing a battery's state of charge using a hydrometer or voltmeter. Visit your nearest Marshall stockistfor a professional battery check A battery must also be scrutinised for any physical condition which may reduce battery life and impede starting performance such as broken or damaged posts and leaks to the battery case or lid. In addition, some electrolysis of the water in the electrolyte occurs breaking it down into its component gases: hydrogen and oxygenīatteries must be subjected to regular testing to ensure their starting capacity is maintained at an optimum performance level. The water produced during discharge is consumed returning the lead to its original strength. During recharge, the electrodes are converted back to lead dioxide and lead. Water is produced and dilutes the strength of the acid. The chemical reaction that takes place during discharge converts both the positive electrode and the negative electrode to lead sulphate. The cell can be recharged by forcing electrical current back through the cell in the reverse direction. The battery becomes discharged (or flat) when no more current flows through the cell. The liquid solution is called an electrolyte which consists of a diluted solution of sulphuric acid. The positive and negative plates are always separated by a separator to the plated, they do not make contact and self discharge. Two or more cells connected together are called a battery. When a positive and a negative electrode are combined (but not touching), they are referred to as a cell. Pure lead is used for the negative electrode or plate and a lead dioxide paste is used for the positive electrode. The different metals are referred to as electrodes. When the metals are connected together above the liquid, electrical current flows through the connection. Electrical current is generated when two different metals are placed separately in a liquid capable of conducting electricity. This stabilising effect also protects a vehicle’s electrical system by smoothing out sudden high voltages which can damage electrical components.īatteries produce their power through a chemical reaction which is released when a load such as a globe, starter motor or electric fan is connected to the battery. Between low engine speeds and when accessory load is greater at higher running speeds, a battery makes up the difference by stabilising the alternator output. It also supplies power to accessories such as lights, fans and radio when the engine is not running. The automotive battery provides starting power for the engine. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
